Inguinal hernia
Frequency:
It ‘s a very common disease. It is estimated that in USA every year 800,000 patients undergo inguinal hernioplasty. Inguinal hernia affects almost exclusively men, from birth to adulthood. Only 3% of women are affected by this pathology.
Pathogenesis:

The book on the genesis of inguinal hernia written by Prof. Amato https://link.springer.com/book/10.1007/978-3-030-95224-2
For over two decades, Professor Amato has undertaken an in-depth research activity on the physiopathology of the inguinal region aimed at establishing the genesis of the disease caused by visceral protrusion. These studies, conducted on patients and cadavers over the years, have produced a wealth of scientific evidence, subsequently published in a long series of scientific articles in the most prestigious international journals. During these studies, the presence of degenerative alterations in the tissues of the inguinal region was observed in individuals with hernias. These degenerative lesions, of a type consistent with damage from chronic compression, affect all inguinal structures, including muscles, arteries, veins, and nerves. (1-6) Thanks to the scientific findings highlighted by Professor Amato, we can now assert that inguinal hernia is a degenerative disease affecting the tissues of the inguinal region, caused by the continuous orthostatic impact of abdominal viscera on the inguinal region.
CLASSIFICATION OF INGUINAL HERNIAS:
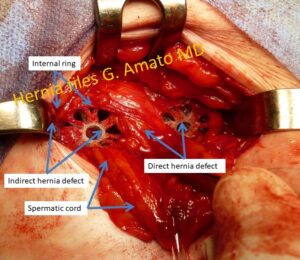
- Indirect inguinal hernia.Is the most frequent protrusion type. The sac erniario protrudes from the lateral inguinal fossa trough the internal inguinal orifice (deep ring). It can be accompanied by preperitoneal lipomas which are relatively frequent.
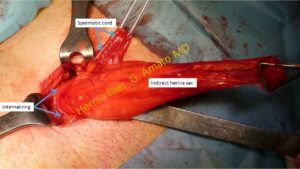
Indirect inguinal hernia protruding through the internal inguinal ring (deep ring) in the lateral inguinal fossa
- Congenital inguinal hernia. It’s always an indirect hernia because it protrudes through the internal inguinal ring. It’s caused by the failure of obliteration after birth of the processus vaginalis of the testicle. As a result, the herniated organs come into direct contact with the testicle. Typically, these hernias become evident a few weeks after birth and are surgically resolved by ligating and cutting the processus vaginalis at the level of the internal inguinal ring without using a prosthesis. In children, these hernia types can easily lead to strangulation of the herniated organ, so prompt surgical intervention is advisable after diagnosis. In adults, these hernias are less common and may only be diagnosed during surgical procedures.
- Direct inguinal hernia. This hernia type protrudes though the fossa inguinalis media. Represent the most frequent type of inguinal protrusion after the indirect hernia
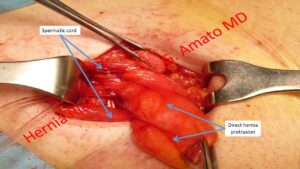
A direct hernia protrudes from the medial inguinal fossa
- Hernia of the sopravescical fossa.
Protrudes from the fossa sopravescicalis, the most medial of the three inguinal fossae. Its outline resembles a diverticular outpouching heading toward the inguinal canal from medial to the external inguinal orifice. Is often mistaken with direct hernia, also by skilled surgeons. Compared to other hernia types results more prone to strangulation because tightened at its basis by a stiffened stricture made up by the medial umbilical fold and the inguinal falx. This hernia type is known for a high tendency to incarceration andstrangulation.
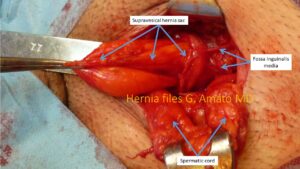
The typical infundibular outline of the hernia of the supravesical fossa
- Combined inguinal hernia.These hernias are typically of large dimensions and result from the confluence of hernia protrusions involving more than one inguinal fossae. Can be categorized into two types: bicomponent and tricomponent combined hernias. Bicomponent combined hernias involve protrusions that affect two adjacent inguinal fossae, either direct and indirect (medial and lateral fossa) or direct and supravesical (medial and supravesical fossa) hernias, which merge into a single protrusion. Tricomponent combined hernias, on the other hand, result from the complete dissolution of the inguinal floor caused by the degeneration induced by the disease. These protrusion types involve all three inguinal fossae: supravesical, medial, and lateral. Combined hernias are the most challenging to treat and have the highest recurrence rate.
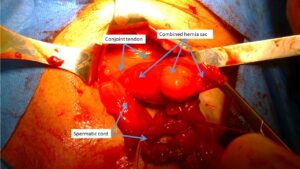
Bicomponent combined hernia protruding from the lateral and medial inguinal fossae
- Inguinoscrotal hernia.This protrusion type occurs when a hernia (direct, indirect, or combined) extends into the scrotum and comes into contact with the tunica vaginalis of the testicle. Typically, it is a large hernia and can potentially cause compressive damage to the testicle, often indicated by the presence of a serous fluid in the testicular sac (hydrocele). Strangulation of this type of hernia is common. Therefore, it is advisable to undergo surgery before the condition further complicates.
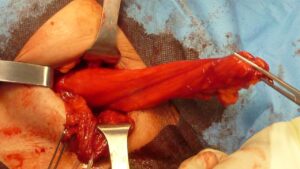
Large hernia sac of inguinoscrotal protrusion after dissection of adhesion bridles
- Multiple ipsilateral inguinal hernias. A variant of inguinal hernias, relatively common, affecting approximately 12% of all herniated patients, is characterized by the presence of multiple separate protrusions arising in the same groin. These hernias can bemultiple indirect (two or more hernia sacs protruding from the internal inguinal ring), multiple ipsilateral (combining indirect and direct, direct and supravesical, or indirect and supravesical hernias), or even triple: three distinct hernias, protruding separately from the three inguinal fossae – supravesical, medial, and lateral. This is a particularly tricky situation because sometimes the protrusions are small and well-hidden among the fatty tissue and adhesions often present in the herniated groin. If not identified during the initial surgery, these hidden hernias can quickly increase in size and cause the same symptoms as the previously operated hernia, leading to pain and discomfort. In such cases, surgical re-intervention may be necessary. Some recurrent hernias are thought to be multiple ipsilateral hernias “forgotten” during the initial procedure.
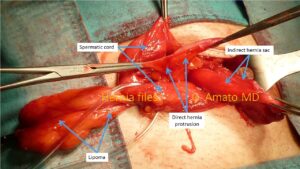
Multiple ipsilateral inguinal hernia composed by an indirect hernia sac, a direct hernia protrusion and one lipoma
Treatment
The primary treatment for an inguinal hernia is surgery, and it should be scheduled before the hernia enlarges. As the hernia grows in size, the risk of complications, such as obstruction or strangulation, increases. These complications are quite dangerous and affect around 10% of patients. In cases of strangulation, the herniated contents can become trapped within the hernia sac, and if not manually reduced back into the abdominal cavity promptly, it can lead to intestinal necrosis, resulting in peritonitis. In these situations, emergency surgery is necessary but carries a high risk of complications.
Surgical techniques and materials
Throughout the years, various techniques have been developed for hernia repair. The oldest technique, practiced until the end of the last century, was the Bassini repair technique, developed by Edoardo Bassini, the director of the surgical clinic at the University of Padua. In 1884, he introduced this technique for inguinal hernia repair, involving the reconstruction of inguinal myotendineal structures using sutures. Several variations of this “pure tissue repair” procedure have emerged over the years, including the Shouldice and McVay techniques.
In 1960, with Usher being the first to use a synthetic material to cover the hernia opening, the era of hernia repair with biocompatible implants began. Lichtenstein further improved the prosthetic hernia repair procedure by standardizing the technique and securing the mesh with sutures. Another innovative contribution was made by Trabucco, who not only covered the hernia opening with a pre-shaped implant without sutures but also introduced a synthetic plug into it the defect. However, it’s important to note that all these techniques were developed without a comprehensive understanding of the causes of hernia genesis, which, as mentioned, at that time was not fully known. Consequently, these methods are not consistent with the pathogenesis of the disease.
Most of the prostheses used today are primarily made of polypropylene. They are typically flat and aim to reinforce the inguinal region by inducing an inflammatory response that leads to the formation of a fibrotic scar plate. After the initial six months of implantation, the prosthesis tends to shrink, losing up to 30% of its surface. In the case of plugs, the shrinkage can be as high as 70%. (1 – 6) At this regard, the concept of cure with flat mesh deployed over the defect to reinforce the groin do not consider the physiology of the inguinal region and often require implant fixation with sutures or similar means, which fully disrespect the dynamics of the abdominal wall.
All the hernia implants used until now are static and are placed by the surgeon in one of the body’s most mobile areas. These prostheses are often fastened in various ways to the inguinal structures. Since they are fixated, they cannot cope with the movement of the inguinal muscles, which impairs the natural dynamics of the groin. The poor quality of tissue ingrowth, resulting in a stiff and shrunken scar plate, can lead to patient discomfort and, in some cases, even chronic pain. Recent scientific studies show that after years of inguinal hernia repair with conventional meshes, from 15 to 20% of patients experience chronic pain. (7 – 12) While this percentage may seem low at first glance, it translates to over 120,000 patients suffering from chronic pain out of the 800,000 inguinal hernia repairs performed each year in the USA. This means enduring, permanent pain that upset life of patients.
Furthermore, other complications can arise due to non-physiological surgical maneuvers, such as mesh fixation, which limits muscle mobility and can tear inguinal tissue. These complications may include bleeding, hematoma, infection, mesh displacement, traumatic injuries to the spermatic cord structures, and recurrence. Additionally, the suboptimal tissue integration with static meshes can result in discomfort, which can have a severe impact on a patient’s quality of life.
The 3D dynamic regenerative scaffold ProFlor: the new frontier in inguinal hernia repair.
The treatment concept for inguinal hernia repair with the 3D dynamic regenerative scaffold ProFlor: the video shows the revolutionary features of the device
https://youtu.be/bB4wPwfIhsE

The 3D dynamic regenerative scaffold ProFlor
Over the years, Professor Giuseppe Amato has delved into the study of the genesis of hernia disease. Scientific studies in recent decades have focused on the effects of inguinal hernias without investigating the underlying causes that lead to the onset of the disease and its associated symptoms. This has resulted in the proliferation of various types of prostheses, all designed to act as a “patch,” essentially covering an area vulnerable to visceral protrusion.
Professor Amato’s studies, on the other hand, highlight tissue degeneration in a portion of the inguinal region and demonstrate how this degeneration is the root cause of the pathology. These studies, conducted over several years, have also been carried out in collaboration with the Pathological Anatomy Institute at the University of Trieste, directed by Professor Furio Silvestri. (13-21)
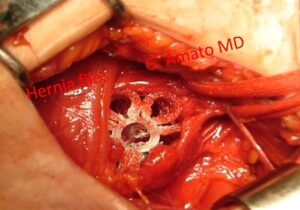
A 25 mm sized 3D dynamic regenerative scaffold ProFlor occupies an indirect inguinal hernia defect in a female patient
Working on the concept of tissue regeneration as a solution to the problem, Professor Amato has developed a 3D dynamic ProFlor scaffold that, when inserted into the hernia defect, obliterates it. Furthermore, unlike conventional prostheses, it harmoniously follows the movements of the surrounding structures and stimulates tissue regeneration. Despite being made of the same material as static prostheses (polypropylene), it’s the unique structural design of the 3D dynamic ProFlor scaffold that allows it to adapt to muscle movements, promoting a different biological response. Instead of the typical growth of a fibrotic scar plate seen with conventional prostheses, there is a regrowth of the structures that characterize the inguinal region, which have been degenerated by hernia pathology. Experimental studies validating these findings have been scientifically confirmed and published. (22-24) This actual regeneration is a result of the continuous movement of the 3D dynamic scaffold, which, when placed within the highly dynamic context of the inguinal region and moving in harmony with the inguinal musculature, attracts growth factors for the regeneration of all components of the inguinal wall. (25-27)
The biological response to hernia devices: A fact to consider
The characteristics of the newly formed tissues inside the 3D dynamic ProFlor scaffold are practically identical to those of the abdominal wall, e.g. muscular structures complete in all their components. The regeneration of new muscle fibers is accompanied by the growth of vascular elements (arteries and veins) and mature nervous structures that support the typical contraction and relaxation activity of muscles normally present in the abdominal wall. In essence, within the context of the dynamic ProFlor 3D prosthesis, there is a true regrowth of all the elements normally present in the inguinal region but lost due to the degenerative injuries of hernia disease. This process of tissue regeneration in the ProFlor dynamic scaffold is activated through the involvement of muscular, vascular, and nervous growth factors, whose presence in the 3D dynamic scaffold has been extensively documented in various post-implantation phases. All of this has been scientifically demonstrated in recent articles (27-31). In particular, the growth of new muscle cells within the 3D dynamic scaffold has been documented and monitored in the short, medium, and long term in another published scientific article (28). Concurrently with the development of muscular structures, another scientific study has demonstrated the presence of complete nervous structures in all their components within the ProFlor, continuing to grow until complete maturation within 6 months of implantation (29).
Another scientific study conducted by Professor Amato compares the biological response in the 3D dynamic ProFlor scaffold to that of conventional prostheses (27).
The results of this research are highly interesting and underscore how, in conventional static prostheses, the incorporating tissue is composed of elements that, over time, lead to the contraction of the prosthesis. The mesh shrinkage is accompanied by chronic inflammation and stiffening due to almost non-elastic fibrous tissue, with rare vascular elements only as support to the inflammation against the implant structure. In contrast, in the dynamic scaffold during the post-implantation phases, there is a gradual growth of new muscles, arteries, veins, and nerves within a context of loose and well-hydrated connective tissue. In practice, all specialized tissue elements typical of the inguinal wall are present, and after a few months, they take on the characteristics of mature cellular elements that are identical to normal tissue.
These significant differences in biological response between the two types of devices should prompt patients who are considering hernioplasty to think carefully about their choice.
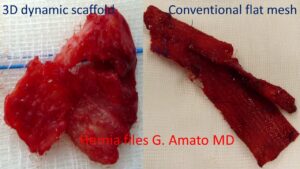
Differences in the biological response between the 3D ProFlor scaffold, which becomes integrated with fleshy tissue, and the conventional flat prosthesis, which is deformed by rigid scar tissue.
In the photo here on the side, a clear contrast is shown in the biological response between the 3D dynamic ProFlor scaffold and a conventional static prosthesis. The photos depict a biopsy of the 3D dynamic scaffold ProFlor and an explanted prosthesis due to recurrence, both excised 8 months after implantation. The structure of the ProFlor scaffold is no longer recognizable because it has been incorporated by fleshy tissue. In contrast, the static conventional prosthesis appears as a shapeless mass of folded and stiffened tissue, surrounded by scar tissue.
The difference is significant: regenerated tissue versus a piece of hardened fabric!
The decision of whether to have a device implanted in their body for hernia repair that, after a few months, will result in an amorphous conglomerate of plastic material mixed with scar tissue infiltrated by an inflammatory response rests with the patients. Alternatively, they can opt for a regenerative scaffold that promotes a probiotic response leading to the growth of vital and well-vascularized fleshy tissue. This is ProFlor, the sole 3D regenerative scaffold that re-establish the integrity of the inguinal barrier.
THE PROFLOR TECHNIQUE FOR INGUINAL HERNIA REPAIR: OPEN AND LAPAROSCOPIC
https://youtu.be/OKV-fglkBN4
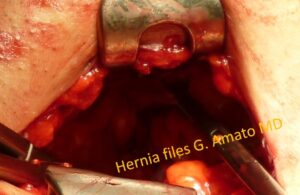
The inguinal floor, which has been completely demolished by a tricomponent combined hernia protrusion, is reduced using forceps in the abdominal cavity.
The surgical technique with the 3D dynamic ProFlor scaffold can be carried our in both open anterior and laparoscopic procedures. In the case of the open technique, the surgery is performed under local anesthesia in almost all cases, and patients can be discharged a few hours after the operation (day surgery). (33-35) The patient preparation and the parts of the procedure prior to the placement of the prosthesis follow the surgeon’s preferred routine. In the case of bilateral hernias or recurrent hernias that have already been operated on using the open anterior approach, the laparoscopic ProFlor technique is recommended. It further reduces surgical trauma and postoperative complications, facilitating almost immediate patient recovery. (36, 37)
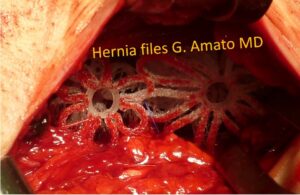
Surgical treatment of large inguinal hernia defect caused by a tricomponent combined hernia protrusion fully obliterated with two 3D dynamic scaffolds ProFlor
The 3D dynamic ProFlor scaffold is inserted into the hernia orifice, where it expands within the defect. Once the hernia orifice is completely occupied, thanks to its centrifugal expansion, the ProFlor scaffold anchors itself autonomously and stably, eliminating the need for the surgeon to use sutures or other fixation methods.
After confirming the correct positioning, the surgeon closes the incision without external stitches, using the total intradermal suture technique with self-locking knots developed by Professor Amato. (38)
Advantages for the patient
The advantages for the patient are:
- Very high postoperative comfort: The 3D dynamic ProFlor scaffold does not induce the formation of fibrotic tissue like conventional prostheses. Instead, it promotes the regeneration of healthy muscular structures with all tissue complements inside. In practice, the patient will have a portion of the abdominal wall that has completely and naturally reformed in place of the hernia defect. In contrast, conventional prostheses that are dynamic passive and motionless produce a poor quality scar plate.
- No need for sutures: The dynamic ProFlor scaffold does not require any form of fixation, eliminating the possibility of painful tissue tension and related complications associated with suture tearing.
- Absence of the typical complications associated with conventional prostheses, such as shrinkage, dislocation, migration, recurrence, discomfort, and chronic pain. (32-37)
Benefits for the surgeon
The advantages for the surgeon are:
- Significant reduction in operative times, primarily due to the rapid placement of the ProFlor scaffold and the fact that, being self-retaining, there is no time wasted on fixation with sutures.
- Easy long-term monitoring of the scaffold placement with a simple ultrasound.
- To date, the recurrence rate ia about 1%. No discomfort or chronic pain have been reported in the operated patients. These are the most feared complications and have the highest impact on the patient’s quality of life.
- The learning curve for the technique is very short, estimated in about 5 procedures.
References
- Lichtenstein IL, Shulman AG, Amid PK, Montllor MM. The tension-free hernioplasty. Am J Surg. 1989 Feb;157(2):188-93. Amid PK. (2004) Lichtenstein tension-free hernioplasty: Its inception, evolution, and principles Hernia 8: 1–7
- Klosterhalfen B, Junge K, Klinge U. The lightweight and large porous mesh concept for hernia repair. Expert Rev Med Devices 2005;2:103–117
- Zhang W, Zhao Y, Shao X, Cheng T, Ji Z, Li J. Long-Term Follow-Up of Lichtenstein Repair of Inguinal Hernia in the Morbid Patients With Self-Gripping Mesh (ProgripTM). Front Surg. 2021 Oct 15;8:748880.
- Wieser M, Rohr S, Romain B. Inguinal hernia repair using the Lichtenstein technique under local anesthesia (with video). J Visc Surg. 2021 Jun;158(3):276-278
- Yu M, Xie WX, Li S, Wang DC, Huang LY. Meta-analysis of mesh-plug repair and Lichtenstein repair in the treatment of primary inguinal hernia. Updates Surg. 2021 Aug;73(4):1297-1306
- Zwaans WAR, Verhagen T, Wouters L, Loos MJA, Roumen RMA, Scheltinga MRM.Groin Pain Characteristics and Recurrence Rates: Three-year Results of a Randomized Controlled Trial Comparing Self-gripping Progrip Mesh and Sutured Polypropylene Mesh for Open Inguinal Hernia Repair. Ann Surg. 2018 Jun;267(6):1028-1033.
- Amid PK. Causes, prevention, and surgical treatment of postherniorrhaphy neuropathic inguinodynia: Triple neurectomy with proximal end implantation. Hernia 2004;8: 343–349]
- Klinge U, Klosterhalfen B, Muller M, Schumpelick V (1999) Foreign body reaction to meshes used for the repair of abdominal wall hernias. Eur J Surg 65:665–673
- Nienhuijs S, Staal E, Strobbe L, Rosman C, Groenewoud H, Bleichrodt R Chronic pain after mesh repair of inguinal hernia: a systematic review. Am J Surg 2007;194:394–400
- O’Dwyer PJ, Kingsnorth AN, Mohillo RG, Small PK, Lammers B, Horeysee G. Randomized clinical trial assessing impact of a lightweight or heavyweight on chronic pain after inguinal hernia repair. Br J Surg 2005; 92:166-70
- Bande D, Moltó L, Pereira JA, Montes A. Chronic pain after groin hernia repair: pain characteristics and impact on quality of life. BMC Surg. 2020 Jul 6;20(1):147.
- Aasvang E, Kehlet H. Surgical management of chronic pain after inguinal hernia repair. Br J Surg 2005; 92: 795–801
- Amato G, Sciacchitano T, Bell SG, Romano G, Cocchiara G, Lo Monte AI, Romano M. Sphincter-like motion following mechanical dilation of the internal inguinal ring during indirect inguinal hernia procedure. Hernia 2009;13:67–72
- Amato G, Marasa L, Sciacchitano T, Bell SG, Romano G, Gioviale MC, Lo Monte AI, Romano M. Histological findings of the internal inguinal ring in patients having indirect inguinal hernia. Hernia 2009;13:259-62
- Amato G, Ober E, Romano G, Salamone G, Agrusa A, Gulotta G, Bussani R. Nerve degeneration in inguinal hernia specimens. Hernia 2011;15:53-58
- Amato G, Romano G, Salamone G, Agrusa A, Saladino VA, Silvestri F, Bussani R. Damage to the vascular structures in inguinal hernia specimens. Hernia 2012;16:63–67
- Amato G, Agrusa A, Romano G, Salamone G, Gulotta G, Silvestri F, Bussani R. Muscle degeneration in inguinal hernia specimens Hernia 2012;16:327-31
- Amato G, Agrusa A, Romano G, Salamone G, Cocorullo G, Mularo S A, Marasa S, Gulotta G. Histological findings in direct inguinal hernia. Hernia 2013;17:757- 63.
- Amato G, Calò PG, Rodolico V, Puleio R, Agrusa A, Gulotta L, Gordini L, Romano G. The Septum Inguinalis: A Clue to Hernia Genesis? J Invest Surg. 2018;31:1-9
- Amato G, Agrusa A, Rodolico V, Caló PG, Puleio R, Romano G. Inguinal Hernia: The Destiny of the Inferior Epigastric Vessels and the Pathogenesis of the Disease. Surg Technol Int. 2020 Mar 18;36.
- Amato, G. (2022). Pathological Anatomy and Histology of the Herniated Groin. In: Inguinal Hernia: Pathophysiology and Genesis of the Disease. Springer, Cham. https://doi.org/10.1007/978-3-030-95224-2_4
- Amato G, Lo Monte AI, Cassata, Damiano G, Romano G, Bussani R. A new prosthetic implant for inguinal hernia repair: its features in a porcine experimental model. Artificial Organs 2011;35(8):E181–E190
- Amato G. Dynamic implant for inguinal hernia repair in porcine experimental model. A feasibility study. Hernia 2009;13:S1-332
- Amato G, Romano G, Rodolico V, Puleio R, Calò PG, Di Buono G, Cicero L, Romano G, Goetze TO, Agrusa A. Dynamic Responsive Inguinal Scaffold Activates Myogenic Growth Factors Finalizing the Regeneration of the Herniated Groin. J Funct Biomater. 2022 Nov 18;13(4):253
- Amato, G.; Puleio, R.; Romano, G.; Calò, P.G.; Di Buono, G.; Cicero, L.; Cassata, G.; Goetze, T.; Buscemi, S.; Agrusa, A Physiologic Cyclical Load on Inguinal Hernia Scaffold ProFlor Turns Biological Response into Tissue Regeneration Biology, 2023, 12(3), 434
- Amato G, Puleio R., Rodolico V, Agrusa A, Calò PG, Di Buono G. Romano G, Goetze T. Enhanced angiogenesis in the 3D dynamic responsive implant for inguinal hernia repair ProFlor Artificial Organs. 2021;45:933–942.
- Amato G, Romano G, Agrusa A, Marasa S, Cocorullo G, Gulotta G, Goetze T, Puleio R. Biologic response of inguinal hernia prosthetics: a comparative study of conventional static meshes versus 3D dynamic implants. Artif Organs 2015;Jan;39(1)
- Amato G, Romano G, Puleio R, Agrusa A, Goetze T, Gulotta E, Gordini L, Erdas E, Calò P. Neomyogenesis in 3D dynamic responsive prosthesis for inguinal hernia repair. Artif. Organs 2018 Dec;42(12):1216-1223
- Amato G, Agrusa A, Puleio R, Calò PG, Goetze T, Romano G Neo-nervegenesis in 3D dynamic responsive implant for inguinal hernia repair. Qualitative study. International Journal of Surgery Volume 76, April 2020, Pages 114-119
- Amato G, Agrusa, Puleio R, Micci G, Cassata G, Cicero L, Di Buono G, Calò PG, Galia M, Romano G. A regenerative 3D scaffold for inguinal hernia repair. MR imaging and histological cross evidence. Qualitative study International Journal of Surgerythis 2021, 96, 106170
- Amato G. Reply to “A commentary on “A regenerative 3D scaffold for inguinal hernia repair. MR imaging and histological cross evidence. Qualitative study” (Int J Surg 2021;96:106170)
- Dynamic autostatic implant for inguinal hernia repair. Early results in an explorative cohort of patients. Hernia 2010;14:S55-S100
- Amato G, Romano G, Agrusa A, Cocorullo G, Gulotta G, Goetze T. Dynamic inguinal hernia repair with a 3d fixation-free and motion-compliant implant: a clinical study. Surg Technol Int. 2014;24:155-65
- Amato G, Agrusa A, Di Buono G, Calò PG, Cassata G, Cicero L, Romano G. Inguinal Hernia: Defect Obliteration with the 3D Dynamic Regenerative Scaffold Proflor™ Surg Technol Int. 2021 May 20;38:199-205
- Amato G, Romano G, Goetze T, Cicero L, Gulotta E, Calò PG, Agrusa A. Fixation free inguinal hernia repair with the 3D dynamic responsive prosthesis ProFlor: Features, procedural steps and long-term results. International Journal of Surgery Open 2019; 21:34-4
- Amato G, Romano G, Calò PG, Di Buono G, Agrusa A. First-in-man permanent laparoscopic fixation free obliteration of inguinal hernia defect with the 3D dynamic responsive implant ProFlor-E®. Case report. Int J Surg Case Rep 2020;77S(Suppl):S2-S7
- Amato G, Agrusa A, Calò PG, Di Buono G, Buscemi S, Cordova A, Zanghì G, Romano G. Fixation free laparoscopic obliteration of inguinal hernia defects with the 3D dynamic responsive scaffold ProFlor. Scientific Reports 2022, 12(1), 18971
- Amato G. Suture intradermiche totali. G Chir. 2010;1/2:48-54